Nature Chemistry
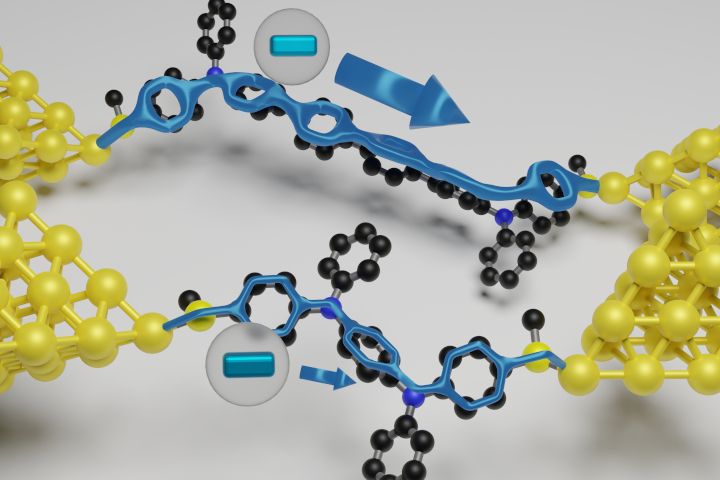
Single-molecule topological insulators are promising candidates as conducting wires over nanometre length scales. A key advantage is their ability to exhibit quasi-metallic transport, in contrast to conjugated molecular wires which typically exhibit a low conductance that decays as the wire length increases. Here, we study a family of oligophenylene-bridged bis(triarylamines) with tunable and stable mono- or di-radicaloid character. These wires can undergo one- and two-electron chemical oxidations to the corresponding mono-cation and di-cation, respectively. We show that the oxidized wires exhibit reversed conductance decay with increasing length, consistent with the expectation for Su–Schrieffer–Heeger-type one-dimensional topological insulators. The 2.6-nm-long di-cation reported here displays a conductance greater than 0.1G0, where G0 is the conductance quantum, a factor of 5,400 greater than the neutral form. The observed conductance–length relationship is similar between the mono-cation and di-cation series. Density functional theory calculations elucidate how the frontier orbitals and delocalization of radicals facilitate the observed non-classical quasi-metallic behaviour.
SFB 1277
Doris Meier
Universität Regensburg
Phone: +49 (0) 941-943 2264
Email: SFB1277.Office@ur.de